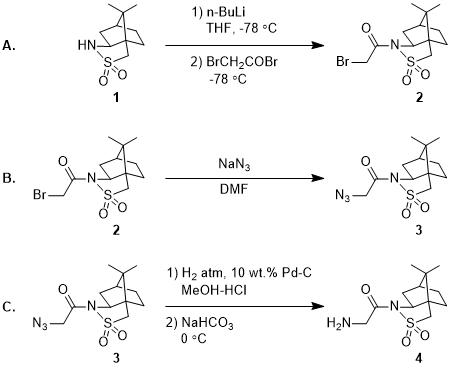
Are n-BuLi/TMEDA-Mediated Arene Ortholithiations Directed? Substituent-Dependent Rates, Substituent-Independent Mechanisms | Journal of the American Chemical Society

n-Butyllithium/N,N,N',N'-Tetramethylethylenediamine-Mediated Ortholithiations of Aryl Oxazolines: Substrate-Dependent Mechanisms | Journal of the American Chemical Society
![Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances](https://www.science.org/cms/asset/860963e6-85a5-48b3-9e47-4a596c258578/abd5290-f1.gif)
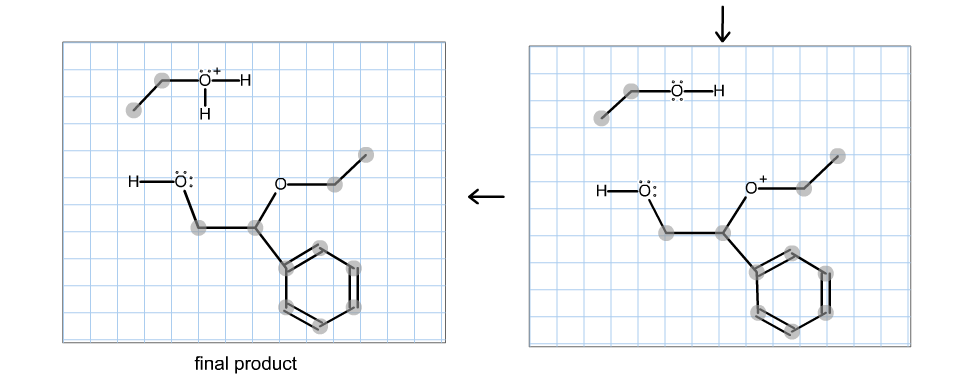
Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances

ortho-Lithiations Reassessed: the Advantages of Deficiency Catalysis in Hydrocarbon Media | Organometallics

Enediolate–Dilithium Amide Mixed Aggregates in the Enantioselective Alkylation of Arylacetic Acids: Structural Studies and a Stereochemical Model | Journal of the American Chemical Society

29 Si CPMAS NMR spectra of (a) carrier C, (b) C treated with BuLi, and... | Download Scientific Diagram
![Concise synthesis of 3-alkylthieno[3,2- b ]thiophenes; building blocks for organic electronic and optoelectronic materials - RSC Advances (RSC Publishing) DOI:10.1039/C9RA08023F Concise synthesis of 3-alkylthieno[3,2- b ]thiophenes; building blocks for organic electronic and optoelectronic materials - RSC Advances (RSC Publishing) DOI:10.1039/C9RA08023F](https://pubs.rsc.org/image/article/2019/RA/c9ra08023f/c9ra08023f-s4_hi-res.gif)
Concise synthesis of 3-alkylthieno[3,2- b ]thiophenes; building blocks for organic electronic and optoelectronic materials - RSC Advances (RSC Publishing) DOI:10.1039/C9RA08023F
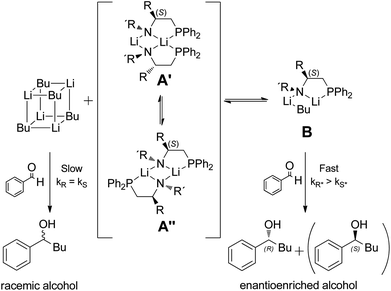
A computational study of the enantioselective addition of n-BuLi to benzaldehyde in the presence of a chiral lithium N,P amide - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB06910E

Metalation of Pyridines with nBuLi−Li−Aminoalkoxide Mixed Aggregates: The Origin of Chemoselectivity | Journal of the American Chemical Society

Two aspects of the desymmetrization of selected prochiral aromatic or vinylic dihalides: enantioselective halogen–lithium exchange and prochiral recognition in chiral liquid crystals - ScienceDirect